‘Pill Mill’ Laws Worked, Cutting Opioid Overprescribing and Deaths
Measures in 11 States Also Reduced Doctor Shopping Without Pushing Patients Into Illicit Drug Use, a New RAND–Penn Study Finds

Measures in 11 States Also Reduced Doctor Shopping Without Pushing Patients Into Illicit Drug Use, a New RAND–Penn Study Finds

Risk-Based Payments May Fail to Lower Low-Value Care, a New Study Shows

New Policy Reduces Housing First While Favoring Enforcement and Involuntary Treatment. That Could Worsen Homelessness, LDI Experts Warn

Follow-Up Texts, Not Paper Flyers, Drove Real Action on Unclaimed Benefits Like Heating, Child Care, Tax Rebates, and Children’s Health

Comparison to Commercial All-Payer Database Documents Wide Discrepancies

These 10 Points Lay Out the Biggest Barriers – From Costs to Complex Rules – and the Reforms Needed to Put More Hospital Care in Homes Nationwide

A New Study Confirms the Outpatient Surge and Suggests That Many Patients Will Be Left Behind

Replacing Flawed Current Payment Methods With the Machine Learning Model “Franklin,” Might Thwart Upcoding and Favorable Enrollee Selection by Medicare Advantage Firms
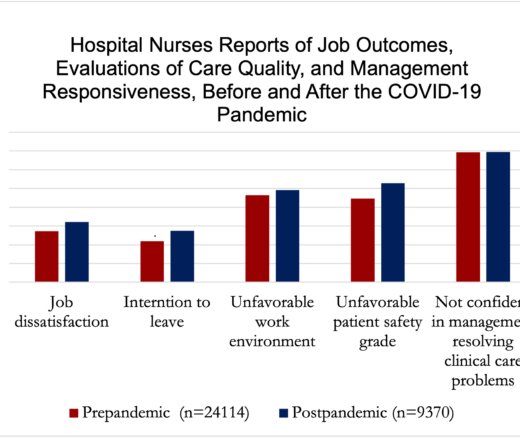
Chart of the Day: National Study Shows Worse Nurse Job Satisfaction and Perceived Lower Care Quality as Staffing Shortages Persist Post-Pandemic
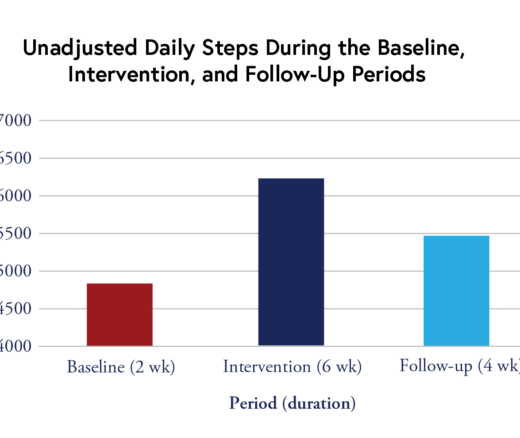
Chart of the Day: Remote Intervention with Gaming Elements Shows Promise, LDI Expert Finds

Federal Officials Factor in Health Gains to Control Spending, LDI Fellow Says

LDI Experts Are Making Postpartum Care Safer With Telehealth, Remote Monitoring, Doulas, and Long-Acting Addiction Care