Blog Post
A Voluntary Program to Encourage Generic Prescribing in Oncology
Study finds little impact on physician behavior, spending
A voluntary UnitedHealthcare program to reduce oncologists’ financial incentives to prescribe brand-name anticancer drugs failed to increase prescribing of generic equivalents. This could be an early warning sign about the limitations of voluntary payment reforms, according to a new study by Laura Yasaitis, Atul Gupta, Justin Bekelman, and colleagues in Health Affairs.
Anticancer drugs have drawn national attention for their high costs and rising share of health care spending. Brand-name anticancer drugs are often much pricier than generics, despite having similar efficacy. Under Medicare’s “buy and bill” payment system, oncologists buy an anticancer drug at one price, and bill at a mark-up price a percentage higher than the original price. Providers have a financial incentive to prescribe brand-name drugs as profits rise with prices. UnitedHealthcare tried to create a win-win situation by increasing providers’ profit margin on generics, while still allowing them to prescribe any available cancer treatment. Even with higher margins, total drug spending would still be lower because the generics are so much cheaper than the branded drugs. Patients would also win as their out-of-pocket spending decreases due to lower copayments.
Providers in the program could switch to an incentivized fee schedule for prescribing any of 12 generic anticancer drugs for adult enrollees with breast, lung, and colorectal cancer. Depending on the drug, the new fee schedule lessened or eliminated the gap in profit margins between generics and brand-name drugs. Over the study period, 695 of 1,905 oncology practices (36%) switched to the incentivized fee schedule. Across all practices, 12,689 patients received an index anticancer regimen, 90% of whom received at least one incentivized anticancer drug in their initial course of treatment. Overall, in the first 30 days of this regimen, mean total spending was $20,624, while mean anticancer drug spending was $10,033 (49%). Mean total out-of-pocket spending was $677.
The program did little to change prescribing patterns or spending in the practices that switched. Adjusted analyses showed that patients in the practice after the switch were no more likely to receive a generic in their first course of treatment than patients before the switch. This experience with the program, which ran from 2007 to 2016, provides some lessons for the development and implementation of future payment reforms.
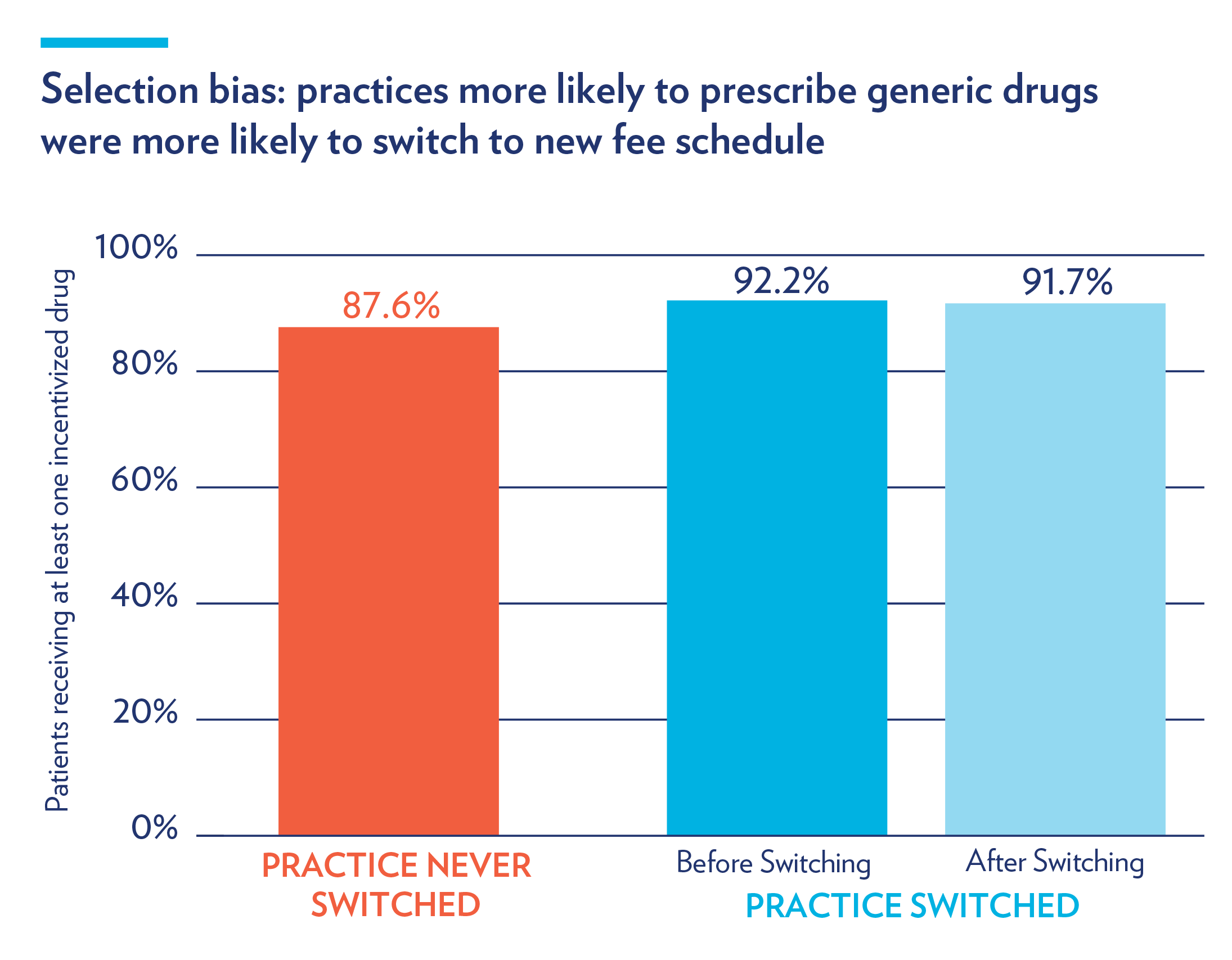
First, oncology practices that opted into the program already had higher rates of generic drug prescribing before switching: compared to non-participating practices, they were more likely to prescribe at least one of the incentivized drugs (92.2% vs. 87.6%). This difference persisted after the switch. This so-called “selection bias,” as illustrated below, is a limitation of other voluntary interventions aimed at changing physician behavior.
At the same time, high rates of generic drug prescribing prior to the program could not explain practices’ lack of response; even those practices with room to increase their generic prescribing exhibited similar patterns to those already prescribing the drugs at a high rate. Finally, the authors hypothesize that in some instances, the incentives may not have been large enough to encourage the switch to generics, although the effectiveness of larger incentives is not necessarily supported in the pay-for-performance literature. Future research could examine what the effects might be if the program was mandatory or implemented among multiple payers, rather than a single insurer.
The study, An Insurer’s Program To Incentivize Generic Oncology Drugs Did Not Alter Treatment Patterns Or Spending On Care, was published on May 6, 2019 in Health Affairs.