
Outpatient Surgeries Still Grew After COVID, Reshaping Care
A New Study Confirms the Outpatient Surge and Suggests That Many Patients Will Be Left Behind
News
Against a backdrop of heightened attention triggered by the FDA’s controversial approval of a new $56,000-a-year Alzheimer’s drug, the University of Pennsylvania’s Leonard Davis Institute of Health Economics (LDI) convened a panel of experts to discuss potential solutions to the national drug pricing crisis.
Opening the September 17 webinar, LDI Executive Director Rachel M. Werner, MD, PhD, noted that prices for the same drugs are much higher in the U.S. than throughout the rest of the world, and a recent survey found that almost a quarter of U.S. adults have trouble paying for their prescription medications.

“This cost is only going up with the prices of brand name drugs increasing at rates that are far above inflation,” Werner said. “And with the recent FDA approval of the new Alzheimer’s disease drug Aduhelm (aducanumab), these issues were brought once again to the forefront, highlighting the complicated interplay between prescription drug prices, access and value.”
Moderated by Werner, the panel included Benedic Ippolito, PhD, a Senior Fellow at the American Enterprise Institute researching health care financing and the pharmaceutical market, Rachel Sachs, JD, MPH, Professor of Law and expert in patent law and drug regulation at the Washington University in St. Louis School of Law; and Mariana Socal, MD, PhD, who studies competition in the pharmaceutical market and access to prescription drugs at the Johns Hopkins Bloomberg School of Public Health.

The Alzheimer’s drug is uniquely interesting because it received accelerated pathway approval even as 10 members of the 11-member FDA scientific advisory committee voted against approval. Three members subsequently resigned in protest because, they said: “There’s no good evidence that the drug works.”
“As a society, we do want to have drugs available in certain situations before we can be 100% sure that they work,” said Socal. “But in this case, the accelerated approval pathway has been utilized for the first time for a drug whose biomarker is not necessarily connected to strong clinical evidence of positive outcomes.”
“This raises the question about how we even reward drugs at this particular stage when we’re not sure they work,” Socal continued. “Would we want to be paying for them at full price?”
But there appears to be little choice because the relationship between pharmaceutical manufacturers, government and consumers is not a standard free market affair. For instance, in the case of the new $56,000 Alzheimer’s drug, Sachs explained that federal regulations leave little wiggle room.

“There might be reason to be concerned that Medicare is going to be held hostage by the makers of this drug because of the way Medicare Part B is structured,” Sachs said. “Medicare can’t say no. Currently, we tell Medicaid ‘You have to cover essentially all FDA-approved drugs.’ We tell Medicare ‘You have to cover drugs that are reasonable and necessary; and we say that drugs that have FDA approval are ‘reasonable and necessary.'”
The industry’s most commonly cited justification for maintaining the world’s highest drug prices is that the revenue supports research and development programs that produce innovative new drug therapies that can save lives in the future. Their argument is that cutting back on today’s high prices and profits will stymie that future innovation.

Ippolito suggested that many people across the country who have health conditions for which there are currently no treatments remain in favor of the high drug prices in hopes that a future innovation will cure their own illnesses.
But countering the idea that high drug prices are the only way to produce the revenue pharmaceutical companies need to innovate, Sachs said it was frustrating that the industry and media constantly cite that option as if it’s the only one.
“There is another direction,” Sachs said. “We can model the increase in new drug sales we get when we expand insurance coverage, either through Medicare Part D, or through the Affordable Care Act and Medicaid expansion. If we give tens of millions of people a new health care benefit, we’re going to increase the uptake of prescription drugs, and that’s going to increase returns to the pharmaceutical industry. Expanding insurance coverage is a drug pricing policy idea that doesn’t get much mention.”
Socal pointed out that when talking about expected new revenue from innovative drugs, most people think about short- and medium-term sales of a patented drug after it is launched in the U.S. market.
“In truth,” said Socal, “the biggest problem is with drugs that have been in the market for a long period of time—more than 10 or 15 years. Everywhere else in the world, there are mechanisms that work over time to bring drug prices down. But in this country, what we see over time is drug prices always going up and up instead of leveling off and declining as in other countries. That’s a problem we need to take care of with our policy.”
Over the last few years, the high level of public frustration and political concerns about the drug pricing situation have spawned a raft of reform proposals on Capitol Hill and in state houses—many of them strongly and surprisingly bipartisan. At the federal level, these include changing the laws to allow the federal government to negotiate drug prices for federal programs, switching to a system that indexes U.S. drug prices to those in other countries—international reference pricing, capping out-of-pocket payments for Medicare Part D recipients, and instituting inflation-based rebates from drug makers beyond those already in place for Medicaid.
Meanwhile, at the state level, some legislators are exploring creation of state drug importation programs to bring in cheaper pharmaceuticals. Others are introducing their own international reference pricing bills. But these proposals have largely languished.
Ippolito, who studies pharmaceutical market regulation and the effect of health care costs on personal finances of Americans, pointed out that even strongly supported bipartisan bills to regulate drug prices in some manner are frequently held back in Congress to be used as leverage in battles over other bills that are less partisan.
“This is not just one political group,” Ippolito said. “Everyone does it. For instance, right now in Congress they have a fix—the Medicare prescription drug benefit. Everyone on the Hill is close to each other politically in terms of normal political negotiation. They could pass that tomorrow and it would help address some of this list of issues we’re discussing here.”
Along with such political inertia, secrecy throughout the interlaced pharmaceutical, insurance companies and pharmacy benefit manager (PBM) industries pose drug pricing reformers’ most formidable challenge: You can’t change what you can’t measure, and the industry has used trade secret laws to keep it this way for drugs. Outside of the list price, very little is known, particularly by consumers.
“As academics and researchers,” said Sachs, “we are frustrated that a lot of this information is secret. It’s really hard to figure out what the numbers are and how we should design policy if we’re estimating something like what Medicaid net prices are, or trying to figure out spending by indication. The industry argues that the net prices paid to different payers are trade secrets and, therefore, cannot be made public. My favorite example is how Pfizer sued the Texas Health and Human Services Commission for disclosing Medicaid prices to its own state lawmakers who were trying to exercise oversight over their own state Medicaid program. Pfizer argued the prices were trade secrets.”
Socal agreed. “Lack of transparency is a big one for patients because typically a patient is required to pay a percentage of the drug price to get their drug,” she said. “That patient will not benefit from the price the insurer actually negotiated for the insurer side. So the patient is required to pay when the drug is dispensed. But that’s too soon to learn the real price. That real net price of the drug gets calculated later on over time. So patients will typically be paying very close to the list price, which is disconnected from the actual price negotiated by the insurer.”
Socal also cited the distorting effect of a widely used rebate system and practices of pharmacy benefit managers, particularly in the competition among branded drugs prescribed through large employer plans.
“The pharmacy benefit manager (PBM) is in charge of negotiating these prices and figuring out which of the branded products is the most convenient and cheapest for the plan,” Socal said. “But, oftentimes, the PBM is making a profit off the price concessions they negotiate. In our research, we’ve seen several examples of situations in which there is a list of cheaper products, but because there’s a profit being made on the price concessions, the most expensive gets treated favorably. Patients end up paying the price—because a decision is made on their behalf by somebody who’s profiting off that decision.”
“A more radical proposal,” said Socal, “would be to just get rid of rebates altogether. Think about a hospital, for example. The hospital is not allowed to send the check back to the insurer and say, ‘Hey, thank you for sending me three people for a colonoscopy here. Here’s a check for five hundred dollars for this volume of patients you sent me.’ That’s illegal in this country, but it is legal for the pharmaceutical manufacturer to send a check and say, ‘Hey, you paid for three units of Humira this month. Here is your rebate.'”
“We could also have a market solution to this problem if more payers were willing to change their contracts with the PBMs and say, ‘No, I’m not paying you a portion of my rebates any more. I’m only going to be paying you fixed fees.’ But at the end of the day, there is an inertia that comes from the models we currently have. Many employers are not willing to risk it. So we need some public policy change in the business model that PBMs use. We need more transparency in how much PBMs make off of every drug.”
In the final question of the day, Werner asked what policy changes offer the best chance of solving the drug pricing conundrum.
Socal, the last speaker, said: “The options are already on the table. Have the HHS Secretary negotiate prices, make pricing information available to all insured individuals, and reference to international drug prices. These policies are very important and would substantially change our ability to negotiate prices going forward. All we have to do now is implement them.”


A New Study Confirms the Outpatient Surge and Suggests That Many Patients Will Be Left Behind

Expert Insights

Replacing Flawed Current Payment Methods With the Machine Learning Model “Franklin,” Might Thwart Upcoding and Favorable Enrollee Selection by Medicare Advantage Firms

A First-of-Its-Kind Pilot Raises Safety Risks and Questions About Who Should Regulate Machines Acting Like Doctors

LDI Fellows Weigh in on the HBO Max Show, Describing Real Challenges From High Patient Bills to Assaults on Nurses in the ER
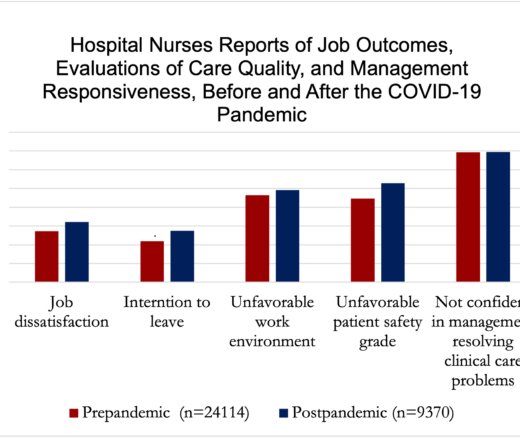
Chart of the Day: National Study Shows Worse Nurse Job Satisfaction and Perceived Lower Care Quality as Staffing Shortages Persist Post-Pandemic
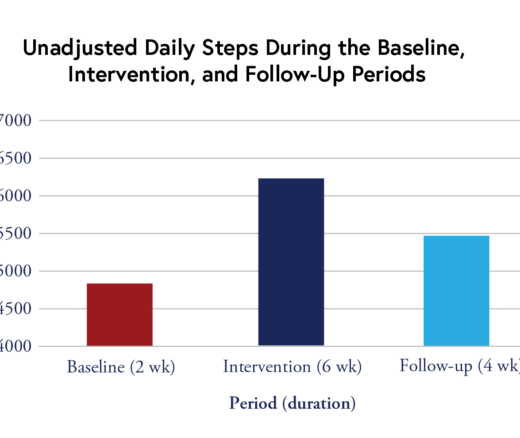
Chart of the Day: Remote Intervention with Gaming Elements Shows Promise, LDI Expert Finds

Cited for Driving Changes in Early Pregnancy Loss Care Guidelines

Penn Researcher Says Mental Illness, Chronic Disease Risks Surge After Release